Discussion :: Chemical Reaction Engineering
-
The rate of the heterogenous catalytic reaction A(g) + B(g)
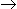 C(g) is given by -rA = k.KA.pA.PB/(1 + KA.PA + Kc.pc), where KA and Kc are the adsorption equilibrium constants. The rate controlling step for this reaction is
C(g) is given by -rA = k.KA.pA.PB/(1 + KA.PA + Kc.pc), where KA and Kc are the adsorption equilibrium constants. The rate controlling step for this reaction is
|
A.
absorption of A.
|
|
B.
surface reaction between absorbed A and absorbed B.
|
|
C.
surface reaction between absorbed A and B in the gas phase.
|
|
D.
surface reaction between A in the gas phase and absorbed B.
|
Answer : Option C
Explanation :
No answer description available for this question.
Be The First To Comment


 Whatsapp
Whatsapp
 Facebook
Facebook

