Discussion :: Chemical Reaction Engineering
-
In the hydrodealkylation of toluene to benzene, the following reactions occur:
C7H8 + H2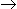 C6H6 + CH4
C6H6 + CH4
2C6H6 C12H10 + H2
C12H10 + H2
Toluene and hydrogen are fed to a reactor in a molar ratio 1:5.80% of the toluene gets converted and the selectivity of benzene(defined as moles of benzene formed/moles of toluene converted) is 90%. The fractional conversion of hydrogen is
Answer : Option B
Explanation :
No answer description available for this question.
Be The First To Comment


 Whatsapp
Whatsapp
 Facebook
Facebook

